Written by Ayan Zeeshan.
Every three seconds, someone in the world develops dementia. Today, more than 55 million people live with dementia globally, a number projected to nearly triple by 2050 as populations age. What was once considered a rare consequence of old age has become one of the defining biomedical challenges of the 21st century.
Overview
Neurodegeneration, the irreversible loss of neurons, disrupts brain-body interactions, causing diseases like Alzheimer’s disease (AD), amyotrophic lateral sclerosis (ALS), Huntington’s disease (HD), multiple sclerosis (MS), and Parkinson’s disease (PD). Factors like aging, genetics, and environment contribute to these disorders. They affect various neurons, leading to speech, movement, sensory, and balance issues. Alzheimer’s features amyloid plaques affecting memory. Unfortunately, as shown by Moriwaka et al ( 1), a gradual rise is observed in developed countries not only in PD, but in other neurodegenerative diseases. We now witness an unprecedented situation in the neurological practice to see so many patients with neurodegenerative diseases which used to be rare.
Core Biological Concepts
The major basic mechanisms leading to neurodegeneration (ND) are considered multifactorial caused by genetic, environmental and endogenous factors related to aging, but their pathogenic role and their basic molecular mechanisms are not fully understood. Neurodegeneration disorders are classified by their protein deposits.
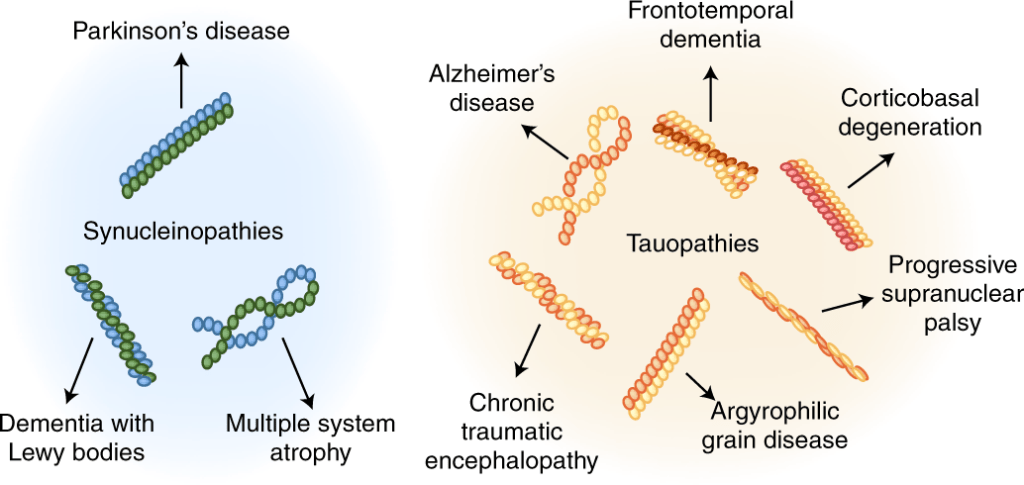
Based on the changes of protein, these disorders are denoted as ‘protein mis-folding’ diseases. Common pathogenic mechanisms underlying many NDDs include:
- Abnormal protein dynamics with misfolding, defective degradation, proteasomal dysfunction and aggregation; often with actions and mutations of molecular chaperones;
- Oxidative stress (OS) and formation of free radicals/reactive oxygen species (ROS);
- Impaired bioenergetics, mitochondrial dysfunctions and DNA damage;
- Fragmentation of neuronal Golgi apparatus;
- Disruption of cellular/axonal transport (4 and 5 may be regarded as secondary effects);
- Dysfunction of neurotrophins (NTFs) and ‘Neuroinflammatory’/neuroimmune processes.
Abnormal interactions between proteins that result in aberrant intra and extracellular deposition of self-aggregating misfolded proteins with formation of high-ordered insoluble fibrils are pathological hallmarks of many, albeit diverse, NDDs. In general, the identity of the underlying protein determines which neurons are affected and, hence, the clinical manifestation of each disease. The same neurodegenerative process and the same mutation in the genes encoding protein constituents may be associated with a variety of clinico-pathological phenotypes, whereas similar or identical phenotypes may be related to different genetic defects, resulting from complex gene–gene and gene-environmental interplay.
Current Scientific Understanding
The integration of molecular biology and biochemistry fosters a holistic understanding of neurodegenerative diseases. Studies have shown that genetic mutations can alter the biochemical
pathways involved in protein degradation, leading to the accumulation of toxic aggregates. In Alzheimer’s disease, the impairment of autophagy and proteasomal degradation systems has been linked to the accumulation of amyloid-beta and tau proteins, highlighting the intricate relationship between genetic factors and cellular biochemistry. Moreover, the advent of high-throughput technologies has revolutionized the study of neurodegenerative diseases by enabling large-scale analyses of gene expression, protein interactions, and metabolic pathways. Techniques such as transcriptomics and proteomics allow researchers to profile the molecular changes that occur in neurodegenerative conditions, providing insights into disease mechanisms and potential therapeutic targets. For example, transcriptomic studies have identified dysregulated gene expression patterns in the brains of individuals with Alzheimer’s disease, revealing alterations in pathways related to synaptic function, inflammation, and lipid metabolism. Such findings underscore the importance of a systems biology approach, where data from various molecular and biochemical analyses can be integrated to construct comprehensive models of neurodegenerative diseases. Despite significant advances in our understanding of neurodegenerative diseases, several challenges remain. The heterogeneity of these conditions complicates the identification of universal biomarkers and therapeutic targets. Individual variability in genetic background, environmental exposures, and lifestyle factors contributes to differences in disease onset and progression. Therefore, personalized medicine approaches that consider the unique molecular and biochemical profiles of individuals may hold promise for improving diagnosis and treatment outcomes. Furthermore, the translation of basic research findings into clinical applications often faces hurdles related to drug development, safety, and efficacy.
Limitations, Unknowns and Research gaps
Two types of lesions have been identified in AD, including amyloid plaques composed of beta amyloid peptides (Aβ), which accumulate abnormally outside the nerve cells, and neurofibrillary tangles due to the hyperphosphorylated tau protein, which accumulates in neurons. AD can be considered a progressive process of biochemical, neurophysiological, neuroanatomical, and cognitive disorders. The initial oligomerization of soluble Aβ in the brain causes localized dysfunctions of dendrites, axonal processes, and synapses. During the past decade, research efforts have focused on soluble Aβ oligomers (AβO), which appear to be a more toxic and disease-relevant form of Aβ. AβO are considered to be pathological agents that appear before the first neuropathological signs of AD. Then, brain lesions gradually form, associated with neuronal loss in certain regions of the brain, but without clinical expression. With time, AD is manifested by loss of memory and cognitive abilities. Many therapeutic strategies have been explored for several decades in clinical trials, but the treatments currently available are primarily treatments of symptoms rather than actual curative therapies. Because of this, attention has turned towards prevention or reducing AD risk. Research has shown that in the world, more than 30% of AD cases could be due to modifiable risk factors, which could provide interesting and promising targets for prevention strategies to reduce the risk of AD-related cognitive decline and perhaps ND in general. Current challenges focus on improving the early detection of the disease at the preclinical stage.
Why does this matters for patients?
Many elderly individuals exhibit mild motor and cognitive alterations reminiscent of those found in neurodegeneration. This observation gave birth to the popular idea that aging might be a “benign” form of neurodegeneration. This idea was supported by the notion, widely accepted until recently, that normal aging, like neurodegeneration, is inevitably associated with neuronal death. From as
early as the 1950s, decreased numbers of neurons in different regions of the brain were reported in
aged humans with no overt neurological or psychiatric conditions (34). Subsequent studies have estimated these losses to be as high as 100,000 neurons per day, which could easily explain the cognitive decline and decrease in brain weight traditionally associated with normal aging (34). However, with the development of more accurate procedures for counting neurons, this view has been modified over the last several years, particularly as stereological procedures for estimating neuron numbers have been applied to aging research. Stereological analysis, on the other hand, describes a methodological approach that provides a three-dimensional interpretation of structures based on observations made on two-dimensional sections, representing the ‘gold standard’ for the unbiased assessment of the structural components of the brain. This means that the strict sampling protocol that stereology requires is often at odds with the need for the rapid and efficient classification of cases, where a balance must be met between diagnostic and research requirements. The inherent benefits of the approach have encouraged researchers to seek out the often subtle changes to the human brain across the various dementing disorders. This review draws together a plethora of diverse stereological studies that have mapped morphological changes in the brain, such as in neuronal populations or vascular integrity, with ante-mortem clinical and/or post-mortem pathological correlates.
Conclusion
Neurodegeneration reflects intertwined cellular stress, inflammation, and protein misfolding that progressively undermine neural networks. Despite expanding knowledge, effective disease modifying therapies remain limited, underscoring the urgency of earlier detection, clearer mechanisms, and patient-centered interventions to preserve cognition.
Citations
1. Kondo, K. (1996). Rising prevalence of neurodegenerative diseases worldwide. Internal medicine, 35(4), 238-238.
2. Jellinger, K. A. (2010). Basic mechanisms of neurodegeneration: a critical update. Journal of cellular and molecular medicine, 14(3), 457-487.
3. Zain, H. (2024). Cross-Disciplinary Approaches to Understanding Neurodegenerative Diseases: Insights from Molecular Biology and Biochemistry. Multidisciplinary Journal of Biochemistry Technology, 1(2), 53-63.
4. Passeri, E., Elkhoury, K., Morsink, M., Broersen, K., Linder, M., Tamayol, A., … & Arab Tehrany, E. (2022). Alzheimer’s disease: treatment strategies and their
limitations. International journal of molecular sciences, 23(22), 13954.
5. Przedborski, S., Vila, M., & Jackson-Lewis, V. (2003). Series Introduction: Neurodegeneration: What is it and where are we?. The Journal of clinical
investigation, 111(1), 3-10. 6. Erskine, D., & Khundakar, A. A. (2016). Stereological approaches to dementia research using human brain tissue. Journal of chemical neuroanatomy, 76, 73-81.
Leave a comment